“While we remain hopeful that the claimed host cells will be deemed patent eligible [in Regenxbio v. Sarepta], the [oral argument] questioning suggests uncertainty about the outcome.”
 In 1980, the Supreme Court decided Diamond v. Chakrabarty, the seminal Section 101 case holding that non-natural, man-made organisms are patent eligible. The Court’s decision paved the way for substantial investment in the then-nascent biotech industry.
In 1980, the Supreme Court decided Diamond v. Chakrabarty, the seminal Section 101 case holding that non-natural, man-made organisms are patent eligible. The Court’s decision paved the way for substantial investment in the then-nascent biotech industry.
Forty-five years later, one would think that there’s little room to debate the patent eligibility of genetically engineered host cells—particularly a host cell that can produce non-native viral plasmids and proteins useful in making life-saving, gene-based medicines. But that debate is front and center in Regenxbio v. Sarepta, a pending appeal at the Federal Circuit that follows more recent Supreme Court and Federal Circuit decisions that have turned Section 101 into a short-cut for bringing patent challenges under Sections 102, 103, and 112.
Regenxbio is appealing a district court’s decision invalidating, as patent ineligible, its patent for a novel, genetically engineered host cell. The Federal Circuit has an opportunity to course-correct in this appeal, which was argued on October 7, 2025. Here, we provide post-argument thoughts about the appeal and why it should largely be controlled by precedent. It goes without saying that reaching the correct outcome is important to maintain the incentives that facilitate advances in genetically engineered technology and medicines.
The Invention and District Court Decision
Regenxbio’s invention is a cultured host cell that contains a recombinant nucleic acid encoded for a specific AAV viral capsid protein and a heterologous non-AAV sequence. In other words, the claimed host cell (such as a bacterial or human cell, for example) is a man-made, genetically engineered cell that can produce plasmids encoding for the viral capsid protein or can produce an engineered capsid with a DNA payload.
The host cell’s novel function is important because it enables the development of life-saving therapies. The viral capsid protein can be used to package and deliver transgenes to patients. The image below, from Regenxbio’s opening brief, depicts one aspect of the patented technology.
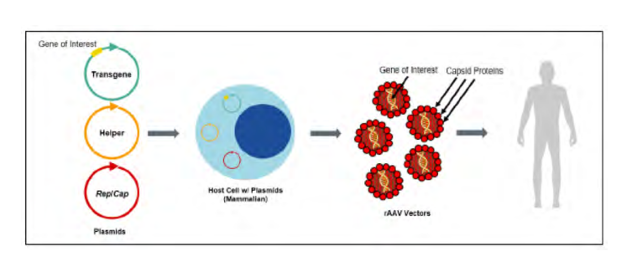
The defendant in this case is accused of infringing the patented technology. According to the complaint, Sarepta developed its gene therapy product, ELEVIDYS®, by using recombinant host cells covered by Regenxbio’s patent.
Notably, Sarepta has not presented a non-infringement defense. Nor does the appeal concern whether the patented invention is anticipated or obvious. Instead, Sarepta argued that the genetically engineered host cell is not patent eligible under Section 101. And Sarepta prevailed on that argument in the district court. There, Judge Andrews viewed the patented host cell as similar to “DNA in a test tube.” He also relied heavily on the Supreme Court’s 1948 decision in Funk Brothers Seed Co. v. Kalo Inoculant Co. The outcome is now in the hands of the Federal Circuit.
At Least Three Prior Decisions Support Patent Eligibility of the Claimed Host Cell
In our view, this should have been a very easy case. Three cases resolve the appeal, and all strongly support the conclusion that the claimed genetically engineered host cell is patent eligible.
First, Chakrabarty should control. In Chakrabarty, the invention was a genetically engineered bacterium that was created—through human intervention—to degrade crude oil. To make the engineered bacterium, the scientists transferred naturally occurring DNA plasmids, which encoded for proteins that could degrade hydrocarbons, into a single Pseudomonas bacterium. By doing so, the scientists created a new bacterium from one that, absent human intervention, could not degrade crude oil. The novel, genetically engineered bacterium was made of biological components that separately existed in nature but were combined in a way that created a “new” “composition of matter” having characteristics that were “possessed by no naturally occurring bacteria.”
In some respect, Regenxbio’s genetically engineered host cell is more “man-made” than the bacterium in Chakrabarty. With Chakrabarty’s invention, the scientists combined four naturally occurring plasmids from different bacteria into a single bacterium. There was no creation of a new DNA sequence or plasmid. In contrast, the scientists in the present case created an entirely new DNA sequence and then used that to transform the host cell to create an engineered man-made cell with properties unlike any pre-existing host cell.
Second, the Federal Circuit’s decision in Association for Molecular Pathology v. Myriad Genetics, Inc. is equally controlling. This case was later appealed to the Supreme Court (three times), eventually resulting in the Myriad decision. But the Supreme Court’s ruling left undisturbed other rulings, including that claim 20 was patent eligible.
Claim 20 was directed to a method of (1) growing host cells transformed with an altered BRCA1 gene, (2) determining all growth rates, and (3) comparing the growth rate. Importantly, the panel in AMP unanimously agreed that this claim was patent eligible largely because it made transformed host cells. “Those cells, like the patent-eligible cells in Chakrabarty, are not naturally occurring. Rather, they are derived by altering a cell to include a foreign gene, resulting in a man-made, transformed cell with enhanced function and utility.” The thread of the panel’s reasoning rested on the logical conclusion flowing from Chakrabarty: “The transformed, man-made nature of the underlying subject matter in Claim 20 makes the claim patent eligible.”
As is evident, there is little, if any, daylight between why AMP claim 20 was patent eligible and why Regenxbio’s claimed recombinant host cell should be patent eligible. And because the panel’s ruling in AMP was undisturbed by Myriad, it should remain good law.
Third, Myriad is equally instructive with its holding that cDNA is patent eligible. There, the Court explained that “cDNA does not present the same obstacles to patentability as naturally occurring, isolated DNA segments.” A cDNA is “an exons-only molecule that is not naturally occurring.” The Court explained that, even though the “the nucleotide sequence of cDNA is dictated by nature,” “the lab technician unquestionably creates something new when cDNA is made.” In other words, “cDNA is not a ‘product of nature’ and is patent eligible under § 101” (with the possible exception of a very short DNA sequence that has no introns and therefore “a short strand of cDNA may be indistinguishable from natural DNA”).
Like the cDNA in Myriad, the novel, man-made nucleic acid here—having the specific AAV capsid protein sequence fused with a non-AAV nucleic acid— is not naturally occurring. The scientists have “unquestionably create[d] something new” that is not produced by nature.
The Federal Circuit Oral Argument
The Federal Circuit panel consisted of Judges Dyk, Hughes, and Stoll. We highlight a few argument points of particular interest. For those interested, the audio of the argument is available here.
The panel’s questioning revealed a deep engagement with the above-noted precedent. While we remain hopeful that the claimed host cells will be deemed patent eligible, the questioning suggests uncertainty about the outcome.
First, some questions seemingly recognized that the claimed invention was more than mere isolation. For instance, Judge Stoll noted that, in Myriad, “there was mere isolation,” but in this case, “there’s no dispute that the cultured host cell did not exist in nature.” It was also notable that the panel did not ask any questions about the “DNA in a test tube” analogy—one invoked by Judge Andrews and Sarepta.
Surprisingly, there was little discussion of Mayo and whether the certain limitations should essentially be disregarded as “insignificant, post-solution activity.” The one exchange on this point invoked the ChromaDex case, but Judge Stoll seemingly appreciated that ChromaDex is fundamentally different because the claims in that case “read on milk.” In other words, ChromaDex is an anticipation case, with the claims construed to cover natural milk. And ChromaDex’s anticipation conclusion is fundamentally different from the present case, as Judge Stoll was seemingly suggesting.
Judge Stoll: “Do you have anything like a natural product that these read on?”
Sarepta Counsel: “The naturally occurring sequence is the Rh10 sequence.”
Judge Stoll: “So, your view is that because the language heterologous non-AAV sequence is so broad, we should just act as if that language is not even in the claim?”
Sarepta Counsel: “I don’t think we should act as if the language is not in the claim. But I do think that we ought to take a look at that language and, under Mayo, identify whether or not that recites or identifies any markedly different characteristics beyond just being an isolated DNA.”
From that exchange, it appears that, in Sarepta’s apparent view, the application of Mayo in the context of this claimed invention reduces to the “markedly different” standard of Chakrabarty and Myriad.
Second, the panel discussed the implications of “markedly different structure” versus “markedly different function” and what was required under Chakrabarty and Myriad. Judges Dyk and Stoll, for example, both asked about whether Supreme Court precedent requires the claims to “expressly recite” the markedly different functions of the claimed host cell. Regenxbio counsel correctly (in our view) explained that it does not. Myriad, after all, held the cDNA to be patent eligible without any requirement for the cDNA to have a different function from the naturally occurring gene. We may see a decision grappling with whether a markedly different structure is sufficient under Chakrabarty and Myriad.
Here though, the transformed host cell also satisfies (in our view) the “markedly different function” prong, to the extent it is required. There is no naturally occurring host cell that has the novel AAV and non-AAV sequences and that can perform the functions described, such as producing AAV-gene-containing plasmids or producing the capsid protein itself.
Third, the concern of preemption was front and center, being one of the first issues raised in questioning. Judge Dyk started by noting that “one of the main purposes of 101 is to prevent preemption and to confine the inventor to what was invented.” The preemption concept has been muted in recent case law, but it played a larger role here, perhaps because it involves a gene-based invention. Regenxbio’s briefing detailed why the claimed host does not preempt all uses of the specific AAV gene.
Notably, concerns about preemption are almost always misplaced. Every claim necessarily preempts all uses of the claimed subject matter for a limited time. A patent claim to a new small molecule “preempts” all uses of that small molecule, but only during the patent term. Such broad claims are not incompatible with the goal of promoting progress in the useful arts. Broad claims, when supported by the necessary disclosure, teach other innovators about critical information that can be used in research and development for follow-on innovations. And cross-licensing creates mutually beneficial arrangements for pioneer innovators and follow-on innovators.
More fundamentally, concerns about claim breadth should be addressed by Sections 102, 103, and 112—not Section 101. Judge Stoll, during argument, suggested as much: “I understand your point that it’s broad, which might go more to enablement or obviousness or anticipation, right? I mean, are those defenses being presented in this case?” Sarepta counsel acknowledged that those defenses were “not presented in this case.” And Judge Stoll’s recognition follows directly from the last footnote in Myriad, where Justice Thomas noted that the Court “express[es] no opinion whether cDNA satisfies the other statutory requirements of patentability,” citing Sections 102, 103, and 112, as well as the Government’s brief making the same point.
Finally, the panel appeared unpersuaded by the district court’s reliance on Funk Brothers. Judge Dyk started the questioning by noting that Regenxbio “make[s] a good point that Funk Brothers was different than this case.” That Supreme Court decision has been the subject of much rightful criticism during its 70-plus-year existence. After all, Funk Brothers is better understood as an obviousness decision.
Awaiting the Outcome
It’s been a little over one month from oral argument. If the panel is unanimous, we might see the decision within a few weeks—but longer if there is internal disagreement. Either way, we are confident that the court will reflect on the issues and consider how precedent informs the outcome.
Image Source: Deposit Photos
Author: Feverpitch
Image ID: 3490146

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)


![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2023/09/Cypris-April-26-2026-sidebar-day-of-webinar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2026/04/Tradespace-Apr-28-2026-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2026/04/Virtual-AI-2026-May-18-19-2026-sidebar-700x500-1.jpg)







![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)





Join the Discussion
2 comments so far.
Doreen Trujillo
November 20, 2025 12:29 pmYears ago, I argued an ex parte appeal involving antibody technology. The issue was obviousness, not eligibility. Judge Dyk was on the panel. He asked me what I wanted the Court to do. I said “reverse the Board’s decision.” Judge Dyk said he was not comfortable doing so with this technology. The best I could get was a remand. (The USPTO admitted making a factual error. So the Court remanded, and the USPTO corrected the error. We did not believe it changed the outcome, but the Court had suggested it would, so the applicant did not appeal again.)
Judge Dyk’s comment always troubled me. His view seemed to be to punt it back to the USPTO if he was not comfortable with the technology. I hope he has become more comfortable over the years.
Pro Say
November 17, 2025 06:29 pm“Supreme Court and Federal Circuit decisions that have turned Section 101 into a short-cut for bringing patent challenges under Sections 102, 103, and 112.”
Bingo.