 Gilead Sciences, Inc. (NASDAQ:GILD) of Foster City, CA, is an American biotechnology developer which has pursued a particular focus on anti-viral pharmaceutical medications in recent years. On September 21st, Gilead announced the successful completion of four Phase III clinical trials for the treatment of all six hepatitis C virus (HCV) genotypes with a combination of sofosbuvir (Sovaldi®) and velpatasvir. The HCV treatment has reportedly been granted a breakthrough designation by the U.S. Food and Drug Administration for expedited approval. Gilead is heavily invested into developing treatments for both hepatitis B and C, which together affect about 400 million people worldwide. The successful trials indicate that Gilead may be able to increase its lead over rival drugmaker AbbVie Inc. (NYSE:ABBV), its largest competition in the HCV treatment market. Higher prescriptions for Gilead’s HCV treatments were encouraging price increases for the company’s stock around the end of August.
Gilead Sciences, Inc. (NASDAQ:GILD) of Foster City, CA, is an American biotechnology developer which has pursued a particular focus on anti-viral pharmaceutical medications in recent years. On September 21st, Gilead announced the successful completion of four Phase III clinical trials for the treatment of all six hepatitis C virus (HCV) genotypes with a combination of sofosbuvir (Sovaldi®) and velpatasvir. The HCV treatment has reportedly been granted a breakthrough designation by the U.S. Food and Drug Administration for expedited approval. Gilead is heavily invested into developing treatments for both hepatitis B and C, which together affect about 400 million people worldwide. The successful trials indicate that Gilead may be able to increase its lead over rival drugmaker AbbVie Inc. (NYSE:ABBV), its largest competition in the HCV treatment market. Higher prescriptions for Gilead’s HCV treatments were encouraging price increases for the company’s stock around the end of August.
Near the end of September, Gilead announced more positive news after Europe’s Committee for Medicinal Products for Human Use (CHMP) gave a positive recommendation for Genvoya, a tablet taken once daily for the treatment of HIV-1. The tablet is Gilead’s first  to include tenofovir alafenamide (TAF), a nucleoside reverse transcriptase inhibitor (NRTI) effective for blocking reverse transcriptase, an enzyme which HIV uses to replicate. The positive recommendation could lead to European Commission approval for prescriptions of Genvoya in all 28 member states of the European Union. A recent $10 billion issue of senior notes by Gilead has sparked speculation that the company may be looking to make an acquisition in the biotech sphere.
to include tenofovir alafenamide (TAF), a nucleoside reverse transcriptase inhibitor (NRTI) effective for blocking reverse transcriptase, an enzyme which HIV uses to replicate. The positive recommendation could lead to European Commission approval for prescriptions of Genvoya in all 28 member states of the European Union. A recent $10 billion issue of senior notes by Gilead has sparked speculation that the company may be looking to make an acquisition in the biotech sphere.
Gilead is not a prolific filer of U.S. patent applications, and it failed to place among the top 300 organizations obtaining U.S. patents in 2014, according to statistics reported by the International Property Owners Association. However, we do see that the corporation currently holds 2,131 active patent grants according to data collected by Innography. The text cluster derived from Gilead’s 59 U.S. patents, which it has earned this year, shows R&D into anti-virals applied through therapeutic methods, pharmaceutically acceptable salts and more.
Gilead’s Issued Patents: From Treatments for Heart Health Conditions to HIV Inhibitors
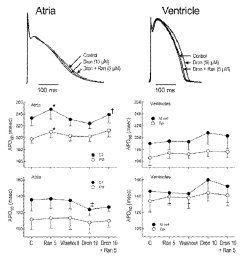 Despite all of the recent focus on Gilead’s anti-viral medications, we noted a trio of patents issued to the company recently by the USPTO to address issues in patient cardiac health. U.S. Patent No. 9056108, issued under the title Method of Treating Atrial Fibrillation, claims a kit containing a first composition which includes dronedarone or a pharmaceutically acceptable salt thereof, and then a second composition which includes ranolazine. This innovation was found to improve the anti-arrhythmic efficacy of dronedarone, which can be used to treat atrial fibrillation but is poorly tolerated by patients in higher doses. Genetic conditions affecting muscle tissues within the heart are addressed by the pharmaceutical invention protected by U.S. Patent No. 9125916, entitled Method of Treating Hypertrophic Cardiomyopathy. It discloses a method for treating hypertrophic cardiomyopathy by administering a specific compound or pharmaceutically acceptable salt to a patient in need thereof. The pharmaceutical medication provides a treatment for hypertrophic cardiomyopathy (HCM), wherein the heart muscle becomes abnormally thick and cannot pump blood as efficiently, by inhibiting the production of sodium which is symptomatic of HCM. A condition that could lead to cardiac arrest and death is the focal point of U.S. Patent No. 9126989, entitled Compound and Method for Treating Long QT Syndrome. It discloses a method of treating LQT syndrome in a patient by administering to the patient an effective amount of a compound, where the effective amount ranges between 0.1 milligrams (mg) and 100 mg. This innovation seeks to provide a treatment for long QT syndrome, a genetic condition which can result in the formation of abnormal sodium channels within the heart which can affect typical electrical signal transmissions.
Despite all of the recent focus on Gilead’s anti-viral medications, we noted a trio of patents issued to the company recently by the USPTO to address issues in patient cardiac health. U.S. Patent No. 9056108, issued under the title Method of Treating Atrial Fibrillation, claims a kit containing a first composition which includes dronedarone or a pharmaceutically acceptable salt thereof, and then a second composition which includes ranolazine. This innovation was found to improve the anti-arrhythmic efficacy of dronedarone, which can be used to treat atrial fibrillation but is poorly tolerated by patients in higher doses. Genetic conditions affecting muscle tissues within the heart are addressed by the pharmaceutical invention protected by U.S. Patent No. 9125916, entitled Method of Treating Hypertrophic Cardiomyopathy. It discloses a method for treating hypertrophic cardiomyopathy by administering a specific compound or pharmaceutically acceptable salt to a patient in need thereof. The pharmaceutical medication provides a treatment for hypertrophic cardiomyopathy (HCM), wherein the heart muscle becomes abnormally thick and cannot pump blood as efficiently, by inhibiting the production of sodium which is symptomatic of HCM. A condition that could lead to cardiac arrest and death is the focal point of U.S. Patent No. 9126989, entitled Compound and Method for Treating Long QT Syndrome. It discloses a method of treating LQT syndrome in a patient by administering to the patient an effective amount of a compound, where the effective amount ranges between 0.1 milligrams (mg) and 100 mg. This innovation seeks to provide a treatment for long QT syndrome, a genetic condition which can result in the formation of abnormal sodium channels within the heart which can affect typical electrical signal transmissions.
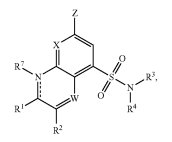 A treatment for viral infections and a wide range of other health conditions which can result in the aberrant expression of genes is detailed within U.S. Patent No. 9108953, entitled Quinoline Derivatives as Bromodomain Inhibitors. This patent claims a compound having a specific formula, one segment thereof selected from the group consisting of hydrogen, halo C1-4 haloalkyl, C3-8 cycloalkyl and oxo. This invention provides a modulator for the bromodomain and extraterminal (BET) family of proteins which can be therapeutically administered to patients suffering from autoimmune disease, cancer, neurodegenerative disease, viral infection or obesity. Treatments for various types of cancers are also protected for Gilead through the issue of U.S. Patent No. 9018221, which is titled Phosphatidylinositol 3-Kinase Inhibitors. It protects a compound of a specific formula having a segment selected from a group including nitrogen and sulfur. This innovation provides an inhibitory agent for phosphatidylinositol 3-kinase (PI3K), an enzyme that mediates proliferative disorders and diseases like pancreatic cancer, bladder cancer, bone cancer or prostate cancer.
A treatment for viral infections and a wide range of other health conditions which can result in the aberrant expression of genes is detailed within U.S. Patent No. 9108953, entitled Quinoline Derivatives as Bromodomain Inhibitors. This patent claims a compound having a specific formula, one segment thereof selected from the group consisting of hydrogen, halo C1-4 haloalkyl, C3-8 cycloalkyl and oxo. This invention provides a modulator for the bromodomain and extraterminal (BET) family of proteins which can be therapeutically administered to patients suffering from autoimmune disease, cancer, neurodegenerative disease, viral infection or obesity. Treatments for various types of cancers are also protected for Gilead through the issue of U.S. Patent No. 9018221, which is titled Phosphatidylinositol 3-Kinase Inhibitors. It protects a compound of a specific formula having a segment selected from a group including nitrogen and sulfur. This innovation provides an inhibitory agent for phosphatidylinositol 3-kinase (PI3K), an enzyme that mediates proliferative disorders and diseases like pancreatic cancer, bladder cancer, bone cancer or prostate cancer.
Gilead has also increased its portfolio of HIV anti-virals with the issue of U.S. Patent No. 8951986, which is titled Salts of HIV Inhibitor Compounds. This patent discloses a citrate salt with an anti-HIV therapeutic agent which is crystalline and can be produced in the form of a tablet. This invention leads to a pharmaceutical tablet with anti-HIV properties that can be consistent in reproduction and therapeutic benefits while also being economically feasible for large-scale commercial production.
Patent Applications of Note: More Tenofovir Medications and Other Anti-Virals
 Our introduction noted an increased use of tenofovir by Gilead in its HIV treatments and we noted a couple of recently filed patent applications discussing novel uses for that compound. U.S. Patent Application No. 20150105350, titled Combination Therapy Comprising Tenofovir Alafenamide Hemifumarate and Cobicistat for Use in the Treatment of Viral Infections, would protect a composition comprising cobicistat and tenofovir alafenamide hemifumarate; the composition would include 50 mg to 500 mg of cobicistat and 3 mg to 40 mg tenofovir alafenamide hemifumarate. The resulting treatment, which can be prescribed to patients with HIV or the hepatitis B virus (HBV), provides effective treatment against those viruses at lower doses of tenofovir by introducing the use of cobicistat. Other novel forms of tenofovir for the treatment of either HIV or HBV are described within U.S. Patent Application No. 20150197535, which is titled Solid Forms of Tenofovir. It would protect a crystal form of a compound characterized by a specific X-ray powder diffraction pattern which is converted into different tenofovir products, such as tenofovir disoproxil (TD) or tenofovir disoproxil fumarate (TDF). This invention results in the production of novel forms of tenofovir in crystal forms for improved processing characteristics.
Our introduction noted an increased use of tenofovir by Gilead in its HIV treatments and we noted a couple of recently filed patent applications discussing novel uses for that compound. U.S. Patent Application No. 20150105350, titled Combination Therapy Comprising Tenofovir Alafenamide Hemifumarate and Cobicistat for Use in the Treatment of Viral Infections, would protect a composition comprising cobicistat and tenofovir alafenamide hemifumarate; the composition would include 50 mg to 500 mg of cobicistat and 3 mg to 40 mg tenofovir alafenamide hemifumarate. The resulting treatment, which can be prescribed to patients with HIV or the hepatitis B virus (HBV), provides effective treatment against those viruses at lower doses of tenofovir by introducing the use of cobicistat. Other novel forms of tenofovir for the treatment of either HIV or HBV are described within U.S. Patent Application No. 20150197535, which is titled Solid Forms of Tenofovir. It would protect a crystal form of a compound characterized by a specific X-ray powder diffraction pattern which is converted into different tenofovir products, such as tenofovir disoproxil (TD) or tenofovir disoproxil fumarate (TDF). This invention results in the production of novel forms of tenofovir in crystal forms for improved processing characteristics.
Patients who have to deal with tuberculosis co-infection on top of HIV may find some relief from the treatment laid out within U.S. Patent Application No. 20150216873, entitled Methods of Treating Patients Co-Infected with HIV and Tuberculosis. The method for treating a patient co-infected with HIV and tuberculosis that would be protected involves administering each of elvitegravir (an HIV integrase inhibitor), an antimycobacterial agent, and a cytochrome P450 inhibitor to the patient; the pharmaceutical administration aims to maintain blood levels of elvitegravir above the minimum concentration necessary for HIV suppression. This innovation was pursued in response to a high incidence of patients having both HIV and tuberculosis, while common TB treatment decreases a patient’s exposure to elvitegravir by increasing cytochrome P450 activity.
 More R&D focus on treatments for HCV can be seen reflected within U.S. Patent Application No. 20150087405, titled Crystal Structure of HCV Polymerase Complexes and Methods of Use. This patent application discloses an isolated polynucleotide comprised of a nucleic acid encoding a polypeptide having a specific amino acid sequence, where the polypeptide has a deletion or substitution of a ? hairpin as compared to a wild-type HCV genotype. This new therapeutic designed to treat HCV infections by helping to inhibit HCV RNA polymerase production.
More R&D focus on treatments for HCV can be seen reflected within U.S. Patent Application No. 20150087405, titled Crystal Structure of HCV Polymerase Complexes and Methods of Use. This patent application discloses an isolated polynucleotide comprised of a nucleic acid encoding a polypeptide having a specific amino acid sequence, where the polypeptide has a deletion or substitution of a ? hairpin as compared to a wild-type HCV genotype. This new therapeutic designed to treat HCV infections by helping to inhibit HCV RNA polymerase production.
Gilead has even been developing treatments for patients suffering from chemical dependencies, as our readers can see in U.S. Patent Application No. 20150250805, entitled Compounds for the Treatment of Addiction. This claims a method of treating chemical dependency on a dopamine-producing agent involving the administration of a compound containing hydrogen for the treatment of addiction to cocaine, opiates, alcohol, nicotine or methamphetamine. This pharmaceutical innovation intends to provide a treatment for alcoholism and other addictions while reducing the tendency for a patient to relapse.
 The treatment of certain cancers like lymphoma and leukemia are featured within U.S. Patent Application No. 20150150881, which is titled Method for Treating Cancers. The method for treating cancer in a subject disclosed here involves administering a therapeutically effective amount of a compound at a dosage level of 800 mg twice daily; the subject in question has a genomic aberration such as a 17p deletion or a NOTCH1 mutation. This pharmaceutical is designed to inhibit the activity of spleen tyrosine kinase, which plays an important role in immunoreceptor signaling, in a human subject having a genetic anomaly causing resistance to treatments for B-cell lymphomas and leukemias.
The treatment of certain cancers like lymphoma and leukemia are featured within U.S. Patent Application No. 20150150881, which is titled Method for Treating Cancers. The method for treating cancer in a subject disclosed here involves administering a therapeutically effective amount of a compound at a dosage level of 800 mg twice daily; the subject in question has a genomic aberration such as a 17p deletion or a NOTCH1 mutation. This pharmaceutical is designed to inhibit the activity of spleen tyrosine kinase, which plays an important role in immunoreceptor signaling, in a human subject having a genetic anomaly causing resistance to treatments for B-cell lymphomas and leukemias.
Finally, we wanted to share a broad spectrum anti-viral medication to treat respiratory viral infections outlined within U.S. Patent Application No. 20150152116, titled Methods and Compounds for Treating Paramyxoviridae Virus Infections. The compound that would be protected here is administered to patients to introduce ribosides, riboside phosphates and prodrugs thereof. This patent application discusses the novel use of certain ribosides showing anti-HCV activity for the treatment of human parainfluenza viruses, which can cause serious lower respiratory tract issues like pneumonia or bronchitis.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[[Advertisement]]](https://ipwatchdog.com/wp-content/uploads/2023/01/2021-Patent-Practice-on-Demand-1.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/UnitedLex-May-2-2024-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Artificial-Intelligence-2024-REPLAY-sidebar-700x500-corrected.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2024/04/Patent-Litigation-Masters-2024-sidebar-700x500-1.jpg)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/2021-Patent-Practice-on-Demand-recorded-Feb-2021-336-x-280.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)






Join the Discussion
No comments yet.