 In late March, it was announced that Teva Pharmaceuticals Industries Ltd. (NYSE:TEVA), an international pharmaceutical developer situated in Petah Tikva, Israel, will spend $3.5 billion to purchase Auspex Pharmaceuticals Inc. (NASDAQ:ASPX), paying $101 per share. The purchase should help improve Teva’s neurological businesses thanks to Auspex’s work in developing treatments for orphan hyperkinetic movement disorders.
In late March, it was announced that Teva Pharmaceuticals Industries Ltd. (NYSE:TEVA), an international pharmaceutical developer situated in Petah Tikva, Israel, will spend $3.5 billion to purchase Auspex Pharmaceuticals Inc. (NASDAQ:ASPX), paying $101 per share. The purchase should help improve Teva’s neurological businesses thanks to Auspex’s work in developing treatments for orphan hyperkinetic movement disorders.
This year the pharmaceutical world has already seen some incredible mergers and acquisitions. This rapid pace of activity represents the highest level of pharmaceutical and biotech takeovers since 2009, according to Bloomberg Business. The week of March 30th started with the announcement of four major pharma or biotech mergers which totaled greater than $17 billion in costs, including the $3.5 billion Teva acquisition of Auspex. One particular deal, the purchase of benefits management company Catamaran Corp. by health insurer UnitedHealth Group Inc., will exceed $12 billion on its own.
Teva is the world’s largest manufacturer of generic pharmaceuticals and fills out more prescriptions for American patients than any other company other than Pfizer. Teva is no stranger to those in the patent world, or to patent litigation specifically. In fact, it seems that Teva is perpetually involved with patent litigation. Earlier this year Teva went all the way to the U.S. Supreme Court to challenge the application of a de novo review standard for claim construction by the United States Court of Appeals for the Federal Circuit. Teva, the patent owner, had sued Sandoz and prevailed at the district court. The Federal Circuit disagreed, ignoring the factual findings reached by the district court. The Supreme Court found that the Federal Circuit
can no longer apply a de novo review standard to claim construction and must review findings of fact relating to extrinsic evidence under the familiar clear error standard.
Company forecasts for revenues during this fiscal year have reached as high as $4.5 billion. Industry insiders had expected that this corporation would engage in more mergers and acquisitions activities for at least a few weeks. Most recently, some critics were speculating that Teva might be getting ready to purchase Mylan, another major generic pharmaceutical manufacturer based in Pennsylvania. Teva’s plans to develop a generic EpiPen this year, which would put them even further into the same market as Mylan. The $3.5 billion purchase of Auspex doesn’t come close to the $45 billion that has been suggested as the price tag for Mylan but this isn’t necessarily the end of Teva’s M&A activities.
Market forecasts are pretty bullish on the market prospects of Teva in the coming months. Some consider the stock to be undervalued as of late March when it had a market capitalization of $53 billion after a 28 percent price increase since last October. Teva CEO Erez Vigodman commented that the Auspex acquisition moves his company more firmly into central nervous system and movement disorder markets, meeting a need that is currently underserved. The research and development produced by Auspex has focused on the development of medicines for movement disorders and orphan diseases such as Huntington’s disease-associated chorea and tardive dyskinesia. Within the Auspex pipeline is a compound known as SD-809 which has been developed into pharmaceutical treatments for involuntary movements associated with neurological disorders. SD-809’s inhibition of the VMAT2 integral membrane protein regulates levels of dopamine neurotransmitters in the brain, helping to control hyperkinetic movement disorders.
[Bio-Pharma]
Notable Auspex Patent Holdings: Deuterium-Enriched Pharmaceutical Compounds for Treating Neurological Disorders and Cardiovascular Disease
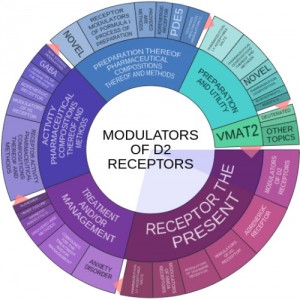 Our research of Auspex’s patents using Innography’s research tools shows us that Auspex holds an IP portfolio of 30 U.S. patent grants and another 193 pending patent applications. As the text cluster here shows that a significant portion of these patent applications are focused on the modulation of D2 receptors, a dopamine receptor, as well as VMAT2.
Our research of Auspex’s patents using Innography’s research tools shows us that Auspex holds an IP portfolio of 30 U.S. patent grants and another 193 pending patent applications. As the text cluster here shows that a significant portion of these patent applications are focused on the modulation of D2 receptors, a dopamine receptor, as well as VMAT2.
Pharmaceutical compositions which have the potential to treat patients suffering from neurological disorders are featured in a number of the patents that were issued to Auspex by the U.S. Patent and Trademark Office. U.S. Patent No. 7872013, entitled Preparation and Utility of Opioid Analgesics, claims a compound containing a mixture of enantiomers, either an individual diastereomer or a mixture of diastereomers and deuterium. The medication is designed to modulate opiate receptors and the N-methyl-D-aspartate (NMDA) receptor to treat and manage pain, anxiety, neurodegeneration or drug dependence without creating additional toxicity through the use of deuterium. The treatment and management of various sleep disorders is the goal of the therapeutic agent protected by U.S. Patent No. 8101633, entitled Preparation and Utility of Substituted Imidazopyridine Compounds with Hypnotic Effects. This patent protects a method of treating a mammal suffering from a condition involving a benzodiazepine receptor through the application of a compound that is enriched with deuterium. This treatment method is capable of addressing a variety of sleeping disorders, from sleep apnea to hypersomnia to sleep sickness, in a way that is less complex and creates fewer adverse effects than conventional treatments. U.S. Patent No. 8962638, which is titled Piperidine Inhibitors of Janus Kinase 3, claims a method of treatment of disorders such as transplant rejection, rheumatoid arthritis, psoriasis, asthma and inflammatory bowel disease through the use of a pharmaceutical compound. The compound is designed to inhibit Janus kinase 3 activity, which could also have beneficial impacts against Alzheimer’s disease and lupus.
Patents issued to Auspex protect plenty of pharmaceutical treatments that aren’t necessarily focused on a patient’s central nervous system. The modulation of receptor activity to treat coronary artery disease or arterial thrombosis is the focus of U.S. Patent No. 8802850, entitled Cyclopropyl Modulators of P2Y12 Receptor. This patent protects another pharmaceutical compound with a different concentration of deuterium than other compounds discussed in the previous paragraph. This invention is intended to improve upon the use of ticagrelor for treating cardiovascular diseases by slowing the metabolization of ticagrelor, which reduces the risk of adverse toxic effects. Coronary disorders as well as asthma and chronic inflammation would be addressed by the compound protected by U.S. Patent No. 8575348, entitled Quinoline Inhibitors of Lipoprotein-Associated Phospholipase A2. This patent protects a deuterium-enriched compound that aids rilapladib, a phospholipase inhibitor that is under investigation for its use atherosclerosis and coronary disorders.
Auspex’s Patent Applications of Note
We took a quick look at the patent applications filed by Auspex that will now become the property of Teva and we found a number of filings related to vesicular monoamine transporter 2, or VMAT2. Treatments for rare and orphan diseases like Huntington’s disease and chronic hyperkinetic movement disorders are discussed within U.S. Patent Application No. 20150080426, titled Benzoquinoline Inhibitors of VMAT 2, protects another kind of deuterium-enriched pharmaceutical compound. The invention provides a benzoquinoline compound which inhibits VMAT2 activity for the treatment of the above diseases as well as hemiballismus, Tourette’s syndrome, depression and cancer. A deuterium-enriched compound enclosed within a pharmaceutically acceptable carrier is at the center of U.S. Patent Application No. 20150080427, titled Benzoquinoline Inhibitors of Vesicular Monoamine Transporter 2. This compound is intended to reduce some of the adverse effects of tetrabenazine, a VMAT2 inhibitor used to treat chronic hyperkinetic movement disorders and which can cause adverse effects such as drowsiness, fatigue, nausea and dizziness. Deuteration which can reduce the negative health impacts caused by tetrabenazine use and drug interactions would also be protected by U.S. Patent Application No. 20150004231, titled Formulations and Pharmacokinetics of Deuterated Benzoquinoline Inhibitors of Vesicular Monoamine Transporter 2. The pharmaceutical composition claimed here is composed of up to 18 percent of d6-tetrabenazine, between 60 percent and 70 percent of mannitol, between 15 percent to 25 percent of microcrystalline cellulose and the rest comprised of polyvinylpyrrolidone, polysorbate and magnesium stearate. Compounds for the treatment of psychotropic disorders are described within U.S. Patent Application No. 20140296339, titled Substituted Phenethylamines with Serotoninergic and/or Norepinephrinergic Activity. The deuterium-enriched compound mitigates some of the shortcomings associated with the use of venlafaxine which is prescribed to treat anxiety disorders, post-traumatic stress disorders, obsessive-compulsive disorder or panic disorder.
We’ll wrap up our research into the patent application holdings that were originally filed by Auspex with a technology that isn’t necessarily in the field of neurological treatments but still involves the use of deuterium. Leukemias and other cancers are the target of the pharmaceutical innovation laid out within U.S. Patent Application No. 20140227260, which is titled Aminopyrimidine Inhibitors of Tyrosine Kinase. The deuterium-enriched compound featured in this patent application is designed to reduce the adverse effects associated with the use of nilotinib, a tyrosine kinase inhibitor prescribed to treat tumors of the colon, prostate, lungs and the breast.

![[IPWatchdog Logo]](https://ipwatchdog.com/wp-content/themes/IPWatchdog%20-%202023/assets/images/temp/logo-small@2x.png)

![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2026/02/Junior-AI-Feb-10-2026-sidebar-CLE-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2026/02/Anaqua-Feb-12-2026-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2026/02/Ankar-AI-Feb-17-2025-sidebar-700x500-1.jpg)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2025/12/LIVE-2026-sidebar-regular-price-700x500-1.jpg)







![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/WEBINAR-336-x-280-px.png)
![[Advertisement]](https://ipwatchdog.com/wp-content/uploads/2021/12/Ad-4-The-Invent-Patent-System™.png)







Join the Discussion
No comments yet.